An acid that ionizes when dissolved in water is called a strong acid. Likewise, a base that ionizes when liquified in water is called a strong base. Both have a few things alike. Both are vital electrolytes that dissociate entirely into ions in water. The ions bring electric fees and also can conduct electrical energy. There are just seven strong acids and also eight-strong bases. They are usually used in chemistry labs, markets, and in daily life [1-4]
Listing of Strong Acids and Bases
Strong Bases
Sodium hydroxide – NaOH
Rubidium hydroxide – RbOH
Caesium hydroxide – CsOH
Strontium hydroxide – Sr(OH)2
Lithium hydroxide – LiOH
Potassium hydroxide – KOH
Calcium hydroxide – Ca(OH)2
Barium hydroxide – Ba(OH)2
Strong Acids
Hydrochloric acid – HCl
Sulfuric acid – H2SO4
Nitric acid – HNO3
Hydrobromic acid – HBr
Hydroiodic acid – HI
Perchloric acid – HClO4
Chloric acid – HClO3
Feature of Strong Acids and also Bases
Strong Acids
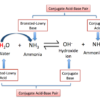
Strong acids are identified by the icon HA, where H implies the hydrogen atom. Then, A is an electronegative atom to which hydrogen is affixed. Strong acids are excellent proton donors and can continue as an aqueous remedy. They are highly destructive compounds that can generate hazardous fumes and create agonizing burns. High concentrations of solid acids ought to be dealt with carefully.
Strong Bases
Strong bases are remarkable proton acceptors and also electron donors. The proton can not remain in a liquid solution for an extended period. Aqueous options of solid bases are unsafe and soapy. Focused services can generate chemical burns.
Ionization of Strong Acids and Bases
Strong Acids
As mentioned, a strong acid can dissociate completely or one hundred per cent in water. The disassociation formula is composed as complies with:
HA (aq.) + H2O (l) → H3O+ (aq.) + A– (aq.).
Right here, water (H2O) as a base responds with the acid (HA), developing hydronium ion (H3O+) and also A–. Hence, A– is the ion that results when the acid sheds a proton.
Why do Strong Acids Entirely Dissociate in Water
To comprehend why strong acids entirely dissociate in water, let us look at the two crucial factors that influence their toughness.
The polarity of the molecule
The toughness of the H-A bond
These two aspects relate to various others. The more polar the particle, the less the electron density around the proton will be. The partial favourable fee on the proton will undoubtedly be high, and the H-A bond will certainly be weak. Because of this, strong acids conveniently dissociate in water.
Strong Bases
Considering that strong bases dissociate completely in water, the disassociation equation can be composed as complies with:
BOH (aq.) → B+ (aq.) + OH– (aq.).
The metal hydroxide (BOH) dissociates wholly right into a metal ion (B+) and hydroxide (OH–). The alkaline earth hydroxides are less soluble in H2O than alkali hydroxides yet are thought about as strong bases.
The strength of Acids and Bases: Ka value
The acid dissociation constant (Ka) indicates how much an acid dissociates in water. Strong acids dissociate entirely into ions and have tremendous Ka values. Another technique of suggesting the toughness of acids is by taking the negative logarithm of Ka, which offers the pKa value.
pKa =– log [Ka]
The lower the pKa, the more influential the acid is.
Strong and Weak Acids.
The hydronium ion (H3O+) has a Ka value of 1 and a pKa value of 0. It is the dividing line between a strong and a weak acid. Simply put, a strong acid is specified as one that ionizes substantially more than H3O+.
The concentration of Strong Acids as well as Bases: pH Value.
The concentration of acids and bases is expressed regarding pH value. Acidic options have a pH near 0, neutral options, such as water, have a pH equal to 7, and standard remedies have a pH near 14. Strong acids have a pH of around 0-1, and strong bases have a pH between 13 and 14. The lower the pH value, the greater the concentration of hydrogen ions in the service.
pH =– log [H+]
Utilizes and Applications of Strong Acids as well as Bases.
Strong Acids.
Hydrochloric acid is used to refine metals, cleanse, and preserve pools. The tummy relies on gastric acid, which is primarily hydrochloric acid.
Sulfuric acid is used in automobile batteries and manufacturing plant foods.
Nitric acid produces fertilizers, makes explosives, and extracts gold.
Strong Base
Salt hydroxide, refer to lye or caustic soda, is used to manufacture soap, cleaning agents, and cleansing options.
Potassium hydroxide, refer to lye or caustic potash, is frequently use to make soaps and cleansing products. It is use in alkaline batteries.
Calcium hydroxide, likewise refer as slaked lime, is use to make concrete and lime water and tidy up spills from solid acids.
Frequently asked questions
Q. 1. Is HF (hydrofluoric acid) a strong acid?
Ans. Hydrofluoric acid (HF) is not a strong acid because it falls short of dissociating in water.
Q. 2. Is NH3 (ammonia) a strong base?
Ans. Ammonia (HN3) is a weak base because it fails to ionize in water completely.